
Dry Etching and Wet Etching - Advantages & Disadvantages of Plasma Etching & Wet Etching
The fundamental process of etching is both simple and ancient: material is removed from a surface using corrosive action. While initially popular centuries ago for engraving copper, etching today is widely used for processing printed circuit boards. In fact, today’s microelectronics industry would be virtually nonexistent without the modern wet and dry etching processes used in electronic device fabrication.
There are two basic etching technologies used today: wet and dry. Both utilize corrosion as the reactive force in the etching process – the difference is wet etching uses chemical solutions while dry etching uses gases. Dry etching is used primarily for circuit-defining steps; wet etching (using chemical baths) is used mainly to clean wafers. Typically, part of the wafer is protected during the etch by an etch-resistant "masking" material, such as photoresist or a hard mask such as silicon nitride.
Here’s an overview of each etching process:
In wet etching, the material is processed using an acidic or basic chemical liquid to remove unwanted material from a surface through a chemical reaction. The acidic or basic chemical liquid used is called the etchant. Wet etching is the simplest process used for etching: wafers are either sprayed or immersed in a chemical bath using solutions such as hydrofluoric acid to etch silicon dioxide over a silicon substrate. It is known as isotropic etching because the corrosive action occurs in all directions simultaneously, at equal rates. Isotropic etching is often used to remove material from a large surface and to clean circuits prior to electroplating.
Wet isotropic etching consists of three general processes: the transport of etchants to the surface of the wafer, a chemical reaction producing soluble by-products, and the movement of reaction products away from the wafer surface. While wet etching uses simple equipment and is relatively fast, it also can be costly, is less precise than dry etching, and produces toxic byproducts that must be properly disposed.
Because dry etching is the primary etching process used in critical manufacturing applications such as those used in microelectronic production, we’re going to take a closer look at this important reactive process.
This process converts gas to plasma in a vacuum environment. Even though dry etching is often used as a synonym for plasma etching, dry etching also includes etching procedures that do not employ RF fields or vacuum.
 The plasma produces high-energy, neutrally-charged free radicals that react on the wafer surface, smashing the plasma ions into the surface to shave material from it. It is similar in some ways to a sandblasting process in which fine-grained sand particles are blasted at high pressure to remove imperfections and discoloration. Dry etching is a more flexible process than wet etching. While dry etching can be isotropic, it can also be anisotropic -- it can achieve more precise, directional material removal as compared to the more general, universal material removal produced by an isotropic process.
The plasma produces high-energy, neutrally-charged free radicals that react on the wafer surface, smashing the plasma ions into the surface to shave material from it. It is similar in some ways to a sandblasting process in which fine-grained sand particles are blasted at high pressure to remove imperfections and discoloration. Dry etching is a more flexible process than wet etching. While dry etching can be isotropic, it can also be anisotropic -- it can achieve more precise, directional material removal as compared to the more general, universal material removal produced by an isotropic process.
Dry etching can etch small patterns with smaller undercut and higher aspect ratios than wet etching. Reactive Ion Etching (RIE), which uses a high pressure plasma as the etchant gas, is widely used for micro-machining. When using a reactive ion etchant, the selected etchant gases must be continuously injected into the vacuum chamber. The radio frequency (RF) power creates plasma which is a mixture of radicals and ions. Reactions of these active high energy species with the surface of wafers create some by-products in the form of gases or other volatile species that will subsequently desorb from the wafer surface and be evacuated through the vacuum exhaust, allowing fresh reactive ion etchants into the vacuum chamber.
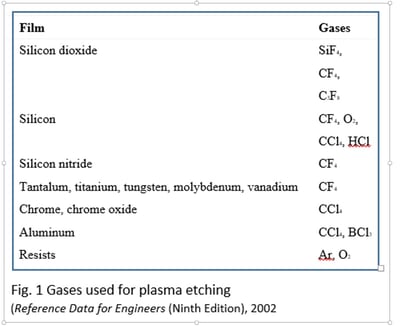 For dry etching in wafer fabrication the reaction chambers vary a lot in their geometrical configuration, pressure range, radio-frequency coupling mode, and gaseous etchants. Fluorine-containing molecules such as CF4, SF6, NF3, and CHF3 are normally employed for plasma-based dry etching of silicon dioxide (Fig. 1). Other gases such as O2, Ar, and He can be added, for example, for heat transfer, plasma stabilization, and enhanced ionization. Typical etch rates for the oxides are in the range of several hundred nanometers per minute.
For dry etching in wafer fabrication the reaction chambers vary a lot in their geometrical configuration, pressure range, radio-frequency coupling mode, and gaseous etchants. Fluorine-containing molecules such as CF4, SF6, NF3, and CHF3 are normally employed for plasma-based dry etching of silicon dioxide (Fig. 1). Other gases such as O2, Ar, and He can be added, for example, for heat transfer, plasma stabilization, and enhanced ionization. Typical etch rates for the oxides are in the range of several hundred nanometers per minute.
Some of the major advantages of dry etching are its capability of automation, reduced material consumption, the ability to use different etch gases with very different process settings in the same tool with little to no hardware change-over time. Also, dry etching can be performed remotely, away from the operator, in a vacuum chamber, reducing health and safety risks. In addition, dry etching chemistry disposal costs less and is easier to dispose of the by-products compared to wet etching.
While the main difference between wet and dry etching is the etching agent utilized, which is either “liquid-phase” in wet etching or “plasma-phase” in dry etching, both entail the removal of layers from a masked surface to achieve three-dimensional nanostructure transfer in a wide range of microelectronic, photonic, and medical applications, to name a few. Which is the most suitable for your application depends on a variety of criteria.
To learn more about etching, check out our eBook titled "Plasma Etching and Cleaning Strategy for Better Product Quality."
Plasma: +1 (248) 761 9253
Distribution: +1 (248) 549 8600
Fax: +1 (248) 549 3533
info@thierry-corp.com